We will look for the best shipping offer to the delivery address. In case of credit card payment, we will send you a payment link in e-mail for the total due amount including the shipping cost, or a proforma invoice with the bank account information in case of wire transfer. Please be aware that the payment link expires after 2-3 days, in case it occurs, please, repeat your order, allowing our staff to generate and send you a new payment link in e-mail. Thank you!
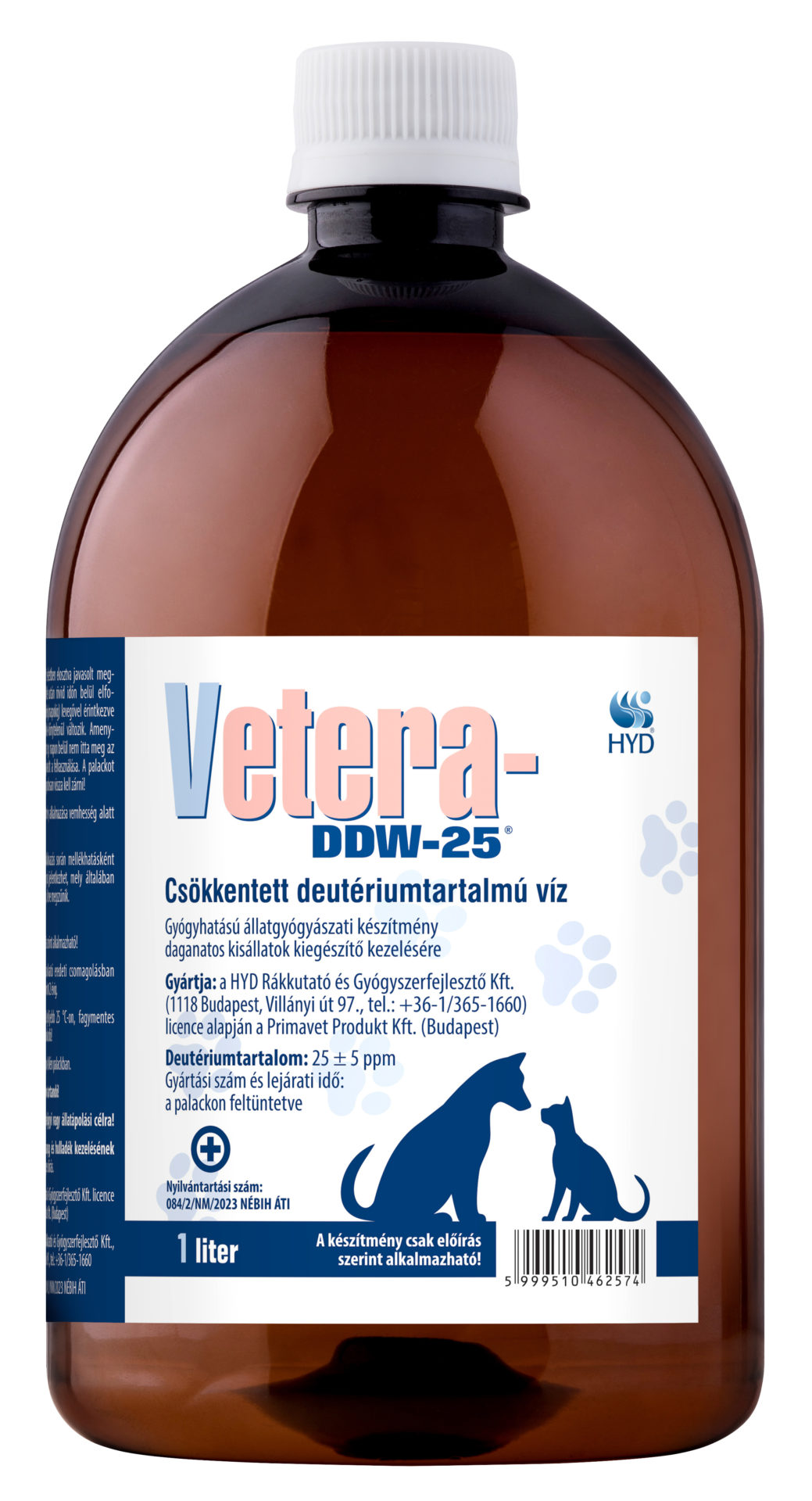
Vetera-DDW-25 Deuterium-depleted water for the adjuvant therapy of neoplastic diseases of household pets
Reg. No.: 084/2/NM/2023 NÉBIH ÁTI
Deuterium content: 25 ppm
Used for the adjuvant therapy of neoplastic diseases of household pets. Main indication of Vetera-DDW-25® is per- and post treatment of surgical interventions or single treatment in case of non-operable tumors.
It is applicable per os (through mouth).
Main advantages of Vetera-DDW-25®:
- high efficacy
- no side effects
- easy to administer
- in-home treatment
Produced by Primavet Produkt Ltd. under the licence of HYD LLC. for Cancer Research and Drug Development.
Recommended daily dose as a function of body mass:
It is advisable to give the daily amount to the animal in two doses. The product should be consumed in a short time after being poured in the bowl; longer contact with the air (for several days) can disadvantageously alter its composition. If the amount poured out was not drunk by the animal within one day, it should not be used any more. The bottle should be firmly closed after every use.
Contraindication: Avoid using the product in pregnant animals.
Side effects: During application, lassitude and drowsiness can appear but these recede or disappear in 2-3 weeks.
Warning: Keep out of reach of children!
Expiry: Unopened bottles can be stored under proper conditions for two years.
Lot number and date of manufacturing: See on the bottle. Do not use the product after the indicated date of expiry.
Storage: Store at 25°C maximum, protected from frost and light.
Packaging: 1.0 L polyethylene bottle
Only for purposes of veterinary medicine or animal care!
Disposal of unused residues and waste: no special requirements.
Step 1: Choose the products you wish to order and put them to the cart. Then, click GO TO ORDER.
Step 2: Enter billing and shipping data, choose payment method, and click SEND ORDER.
Step 3: You will receive a confirmation about your order via e-mail.
Step 4: We will pick the best shipping offer to your address and send proforma invoice via e-mail. (It usually takes 1-2 days to get the delivery charges.)
Step 5: Payment will be done in accordance with the chosen payment option:
By credit card: Proforma invoice will be sent by e-mail with a payment link for credit card payment. Please, click the link, and you will be directed to the SimplePay page, where you can enter your credit card details and complete the process.
Via wire transfer: Proforma invoice will be sent by e-mail with all the bank account information, and the sum total should be transferred.
Step 6: Upon receipt of the payment, the shipment will be started to the delivery address.